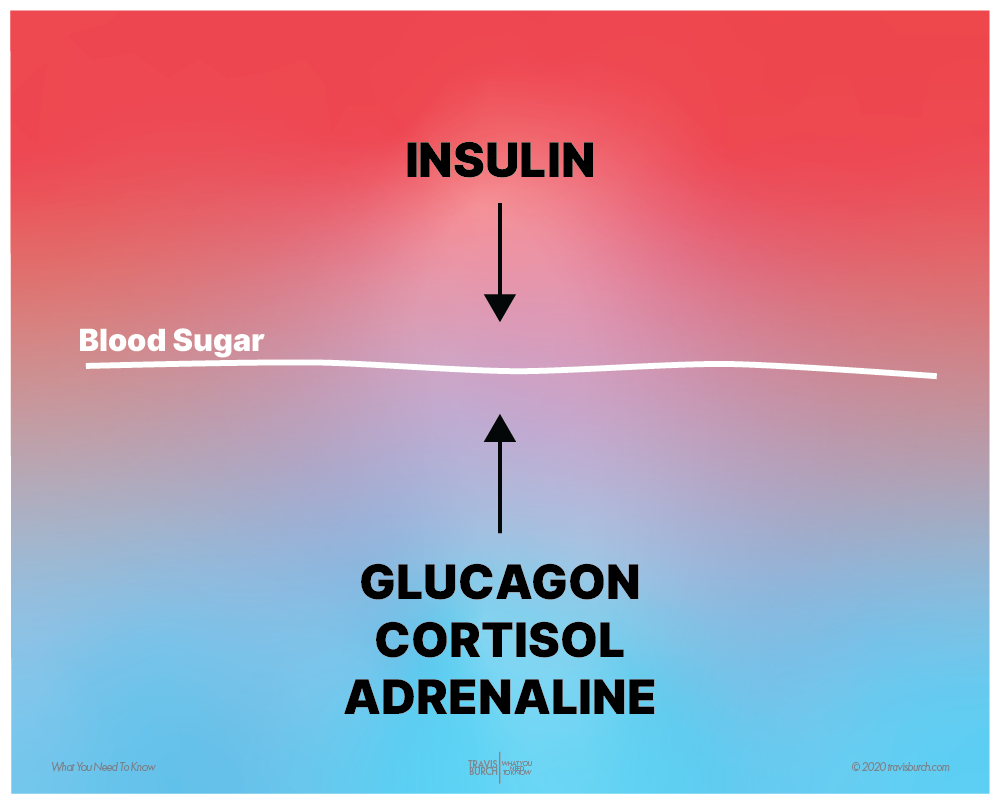
Blood sugar is tightly regulated by two opposing forces: insulin & the stress hormones.
Blood sugar doesn’t stay perfectly balanced all day, though.
After carbohydrates are eaten, they break down into glucose and enter the bloodstream — causing elevated blood glucose.
In response, insulin rises to push it back down to baseline — by driving that glucose into cells. It takes about 2 hours after a meal for insulin to fully lower blood sugar back to baseline levels.
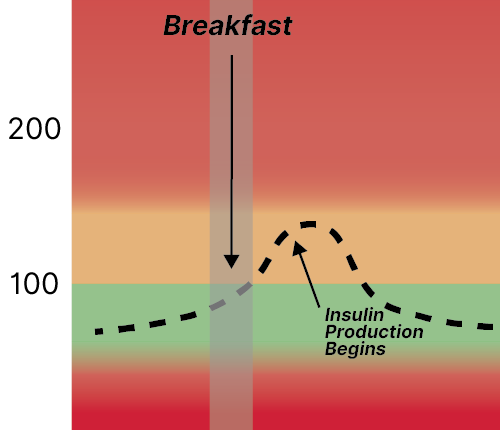
Protein and fat do not spike blood sugar — only carbohydrates. And that’s okay, the pancreas is ready to release insulin after a meal.
Opposing Forces
These hormones (insulin and the stress hormones) are always working in small amounts to keep blood sugar stable — just right.
Think of a faucet with two handles producing lukewarm water:

Balanced blood sugar requires a little insulin and a little stress hormones — at all times.
There’s never a time when any of these hormones should be turned off. They constantly are released in just the right amounts to maintain healthy, stable blood glucose.
What’s happening when blood sugar is unstable?
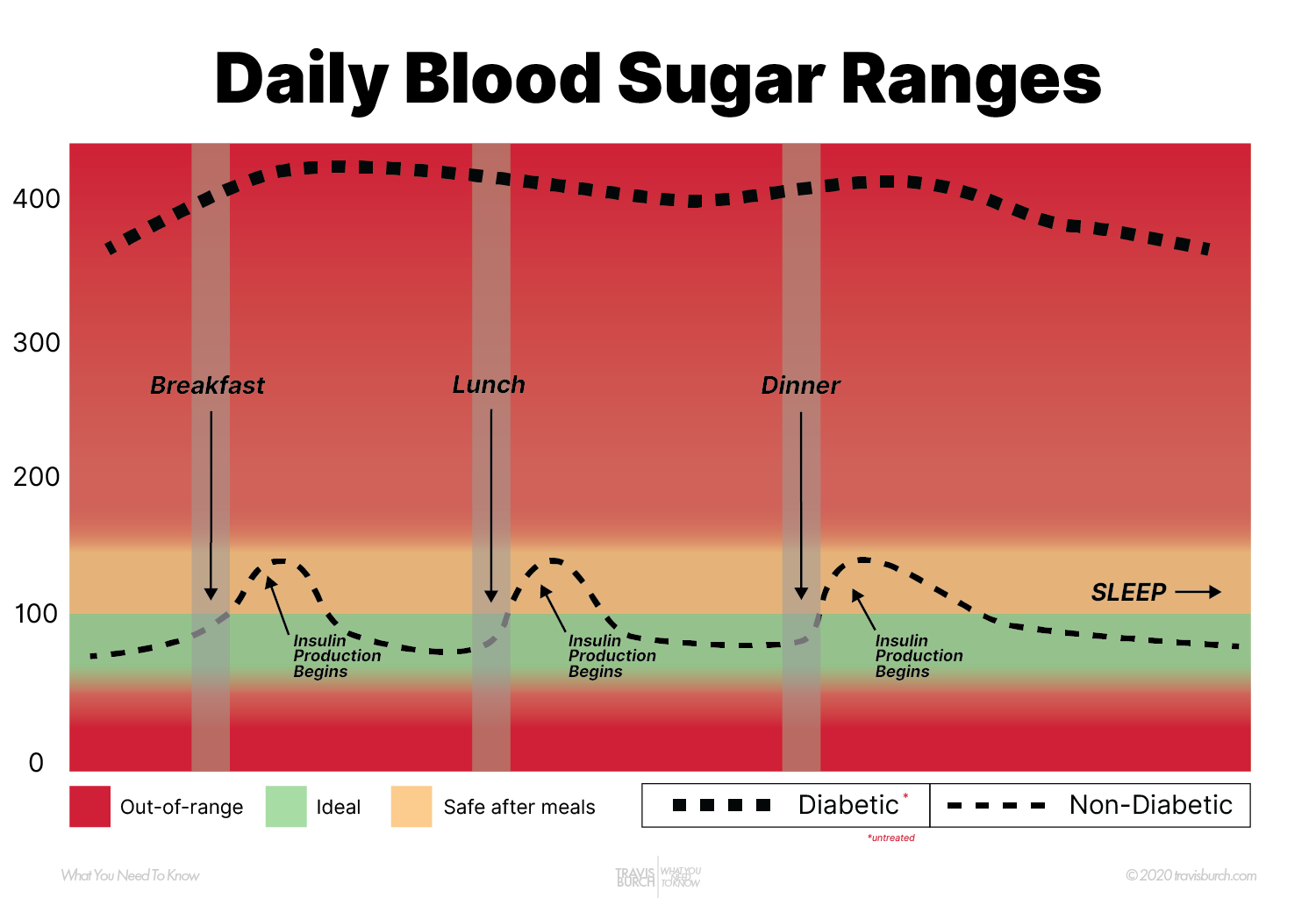
Healthy, stable blood sugar rests between 70-100 (mmol/L).
In diabetes, blood sugar stays high — never falling back to baseline after a meal.
In hypoglycemia, the opposite happens: blood sugar stays low and never gets up to baseline.
There are two reasons blood sugar stays high in diabetes — and both involve insulin:
Healthy Glucose Cycle
In between meals, blood sugar is “just right” — around 90-100 (mmol/L).
“Fasting” blood glucose is measured when you haven’t been eating for 6-8 hours — and it measures how well your body can regulate blood sugar in the absence of meals.
…
When eating carbohydrates, blood sugar rises, possibly up to 250 (mmol/L).
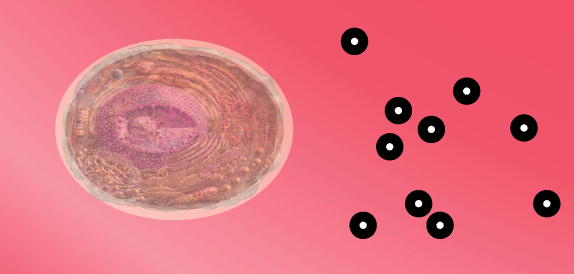
…
Then, the pancreas senses blood sugar and releases insulin.
Insulin attaches to cells, opening the cell membrane. Glucose (and other nutrients) enter the cell.
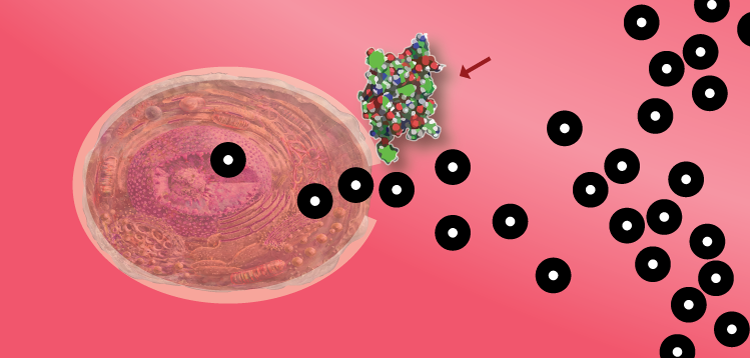
…
Cells then use the glucose for energy — or store it as glycogen to be utilized later.
What Is Diabetes?
Due to either insulin resistance or lack of insulin, blood sugar cannot enter cells (where it’s needed) and leave the blood (where it’s harmful).
Therefore, blood glucose rises and rises — with no solution to counter it.

- Type 1 is less common — it’s when the pancreas has “given out” (usually after years of T2 diabetes).
- Type 2 diabetes is common — it’s merely insulin resistance that has progressed.
“Double diabetes” when both Type 1 and Type 2 symptoms present: poor insulin production (T1) and insulin resistance (T2).

Insulin Resistance
A factor in chronic illness, pre-diabetes, & Type 2 diabetes.
In good health, our cells are sensitive to insulin.
In other words, a little insulin goes a long way.
The pancreas pumps it out, the job is done, and the pancreas rests, unburdened until the next meal.
When cells become resistant to insulin, the pancreas must work harder — pumping out more and more insulin to drive glucose into cells.
In other words, a lot of insulin doesn’t go very far.
This dynamic is a staple of chronic illness and aging — even without a diabetes diagnosis: Nearly everyone who is chronically ill or aging is dealing with some amount of insulin resistance.
Chronic Energy Deficit
Insulin resistance causes cells to become starved for glucose (energy).
Eating more carbohydrates — the normal source of glucose — does not solve the problem: Glucose isn’t entering cells, and instead backs up in the blood.
Why might a cell become resistant to insulin?
Diet and exercise contribute to insulin resistance, but let’s skip those two topics for the moment. Why?
On one hand, there’s controversy and conflicting research about how and which foods affect insulin resistance — while on the other hand, everyone can agree that a little moderate exercise is probably a good idea, generally.
In short, when it comes to insulin sensitivity, 1) diet is a tricky topic (and highly individual), while 2) exercise is a pretty straightforward concept (it’s beneficial when not too stressful).
Therefore, let’s move on to more discussion-worthy causes of insulin resistance.
Inflammation impairs the activity of insulin.
Insulin rises, but cells don’t respond to it — they don’t open up to receive the glucose, because inflammation is in the way.
Nutrients — many of which are also delivered to cells via insulin — too, cannot reach cells.
As a result, cells become starved for energy and deficient in nutrients.
Inflammation plays an important role in the development of IR [insulin resistance].
https://www.hindawi.com/journals/ije/2015/508409/
Energy deficits and nutrient deficiency both lead directly to disease — and can make one feel lethargic and quite unwell every day.
Anything that causes chronic inflammation will lead to insulin resistance.
Worldwide, 3 of 5 people die due to chronic inflammatory diseases like stroke, chronic respiratory diseases, heart disorders, cancer, obesity, and diabetes
https://www.ncbi.nlm.nih.gov/books/NBK493173/
According to this paper, causes of chronic inflammation include:
SOURCE: https://www.ncbi.nlm.nih.gov/books/NBK493173/
To reduce chronic inflammation, consider improving the following components of your health.
Inflammation, interfering with cellular energy production, is probably the essential feature of the things called diabetes.
Ray Peat, PhD
Toxins in the body directly lead to insulin resistance (as well as reduced production of insulin).
In fact, many well-known toxins so directly inhibit glucose metabolism that they are called diabetogens. As carcinogens are to cancer, so diabetogens are to diabetes.
Adding up the numbers shows potentially the whole epidemic is apparently due to the massive increase in body load of toxins.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4991654/
Diabetogens include: BPA, arsenic, dioxins, phthalates, and mold toxins. Endotoxin — produced in the gut by dysbiotic microbes — is another culprit.
Phthalates are used to stabilize fragrances — In one study, reducing use of products with fragrance dramatically lowered phthalate levels in the blood.
These diabetogenic toxins mostly impair insulin sensitivity (T2), but also can directly impede insulin production in the pancreas (T1).
The modern world often considers nutrient deficiency a thing of the past.
This couldn’t be more wrong.
Inflammation of any sort impairs nutrient delivery to cells. When chronic, the inflammatory state results in pernicious nutrient deficiencies in cells.
Absorption through the gut membrane also suffers in an inflammatory state.
Consider 3 of 5 humans die of chronic inflammatory conditions — it’s almost certain that nutrient deficiencies play a role in ongoing illness and aging.
Emerging evidence indicates that micronutrient deficiency could play a significant role in the pathogenesis and progression of many chronic diseases including diabetes mellitus.***
https://www.heighpubs.org/hfns/afns-aid1013.php
*** “…including diabetes mellitus, hypertension, obesity, dyslipidemia, hyperuricemia, kidney disease, cancer, anemia and other cardio-metabolic and neurodegenerative diseases through the induction of Insulin resistance (IR).
Lower levels of potassium have been found to be associated with a higher risk of diabetes in some studies.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3197792/
A large number of nutrients have proven to be depleted in diabetes, including Vitamin A:

Vitamin B3 (niacinamide) is also strongly linked to diabetes and blood sugar management:

Chronic inflammation leads to nutrient deficiency cause each other. Ultimately, both lead to insulin resistance.
When we sleep poorly — or on a late schedule — our glucose metabolism is disrupted.
Sensitivity to insulin is impaired directly, even by a single night of poor sleep.
In fact, one study in dogs found that a single night of poor sleep was more harmful to glucose metabolism than 6 months on a poor diet.
This is why we eat more calories following a bad night of sleep — the body actually requires more food — with a smaller percentage of that food being utilized efficiently by cells.
The circadian timing system is responsible for the coordination of many daily processes, including the daily rhythm in human glucose metabolism.
https://www.ncbi.nlm.nih.gov/pubmed/30531917

If one night of poor sleep can be deeply detrimental to glucose metabolism and also causes insulin resistance. Imagine the effect of long-term sleep disruption.
Pathogens in the body cause an immune response — always.
This immune response means inflammation — which means nutrients can’t be delivered to cells efficiently.
Insulin resistance, inflammation, and nutrient deficiency all cause each other in a pernicious cycle that leads to energy deficit.
When the immune system is compromised — as it is in aging and chronic illness — the body’s microbiome becomes inhabited with opportunistic pathogens in the gut, mouth, nose, ears, skin, and vagina.
“Failure to eliminate infectious organisms including Mycobacterium tuberculosis, protozoa, fungi, and other parasites that can resist host defenses and remain in the tissue for an extended period.”
https://www.ncbi.nlm.nih.gov/books/NBK493173/
A weak microbiome does not control its pathogens.
Pathogens seek to spread around the body, ever-releasing endotoxins, competing for nutrients.
The mere presence of pathogens activates the immune response, causing inflammation system-wide:
- Gut microbes in the gut enter the bloodstream as the gut barrier weakens
- Oral microbes swallowed and enter the bloodstream via weakened gums
- Nasal microbes cause body-wide inflammation and are directly linked many serious diseases
Autoimmunity — another inflammatory scenario — may very likely be triggered by an initial infection that fails to be eradicated fully by the immune system.
Type 1 Diabetes
The pancreas slows production of insulin
Type 1 diabetes is more rare — and more straightforward.
The pancreas just isn’t producing enough, or any, insulin.
Glucose (from carbs) enters the bloodstream — and insulin never arrives to push it into cells.
Why? What causes Type 1 diabetes?
Interestingly, carbohydrates — and sugar — aren’t on this list.
That’s because something else causes Type 1 diabetes — something that attacks the pancreatic cells and prevents them from producing insulin properly.
No amount of sugar in your diet – or anything in your lifestyle – has caused or can cause you to get Type 1 diabetes.
https://www.diabetes.org.uk/guide-to-diabetes/enjoy-food/eating-with-diabetes/diabetes-food-myths/myth-sugar-causes-diabetes
While insulin resistance does occur in Type 1 diabetes — and can reduce the effectiveness of any insulin injections — it is not considered a cause at this time.
So what is considered the primary cause of Type 1 diabetes?
- Viruses
- Toxins
- Autoimmunity
Autoimmunity is where the body’s immune system mistakenly destroys some of its own tissues.
What causes autoimmunity?
Here’s the official medical stance: “The exact cause of autoimmunity is unknown.“
However, there are loads of research implicating infectious pathogens as a primary cause of autoimmunity.
Viruses, bacteria and other infectious pathogens are the major postulated environmental triggers of autoimmunity.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2665673/
Pathogens can release proteins that mimic bodily chemicals — creating a mistaken immune response against host tissues.
We have shown that a bacterium from the gut produces a very close molecular mimic of ubiquitin that gets into our system. It may not only cause the immune system to malfunction, but could be an underlying cause of other human conditions that we don’t yet fully understand.”
Sheila Patrick, professor emeritus, Queen’s University
So we see that autoimmunity is directly caused by the presence of pathogens in the body.
But microbes aren’t only linked to autoimmunity — they’re directly tied to Type 1 diabetes.
Epidemiological evidence suggests that pathogens play a role in development [of T1D]. Many different viruses have been associated with T1D development.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2665673/
Thus, pathogens can cause T1 diabetes directly, or cause autoimmunity — which can develop into T1 diabetes.
Pathogens Also Cause Insulin Resistance?
Pathogens don’t only lead to T1 diabetes, they’re also a primary cause of insulin resistance.
Insulin resistance — the main feature of Type 2 diabetes — develops due to inflammation caused by low-grade pathogenic load..
In fact, higher numbers of pathogens living in the body resulted in a higher incidence of insulin resistance, in this study.
Pathogen burden showed the strongest association with insulin resistance. We hypothesize that exposure to multiple pathogens could cause a chronic low-grade inflammation, resulting in insulin resistance.
https://www.ncbi.nlm.nih.gov/pubmed/16644637
The more pathogens reside the body, the higher inflammation rises.
Higher inflammation means more insulin resistance.
Root Causes Of Both Types Of Diabetes
Thus, we see a strong connection between both types of diabetes — pathogens.
- Pathogens cause insulin resistance (via inflammation)
- Pathogens cause the pancreas to stop producing insulin (either directly or via autoimmunity)
The other main connection between Type 1 & 2 diabetes: toxins.
These diabetogens affect not just insulin resistance, but also directly attack the pancreas’ ability to produce insulin.

What About Genetics?
Diabetes — and insulin resistance — go much deeper than genetics.
In fact, only 10% of those with Type 1 diabetes have a family member with the condition.
We now see that juvenile instances of diabetes — long thought to be genetic — are often preceded by an infection.
When there are genes associated with diabetes (such as: HLA-DQA1, HLA-DQB1, and HLA-DRB1), we see these genes control cell receptors and proteins that are actually critical for the function of the immune system.
In some way, all components of disease manifest as poor immunity to infectious pathogens — including diabetes.







